There are numerous examples of the harsh consequences of inadequate representation in clinical research such as the disregard for biological differences between male and female bodies. Researchers analyzed data from 5000 articles and observed a ...

There are numerous examples of the harsh consequences of inadequate representation in clinical research such as the disregard for biological differences between male and female bodies. Researchers analyzed data from 5000 articles and observed a drug dose gender gap for 86 different medications approved by the FDA, including antidepressants, pain, and cardiovascular medications. The majority of these studies showed that women experience higher relative exposures to the drug, therefore, explaining a higher rate of side effects.
In cancer clinical trials, racial/ethnic minorities have remained persistently underrepresented. While Black Americans make up about 13% of the U.S. population, in breast cancer and prostate cancer clinical trials, Black Americans account for about 9% and 6% of study participants, respectively. Current genomic disparities in the context of this lack of adequate minority participation in clinical trials will lead to major gaps in whom precision medicine services in the U.S. and globally.
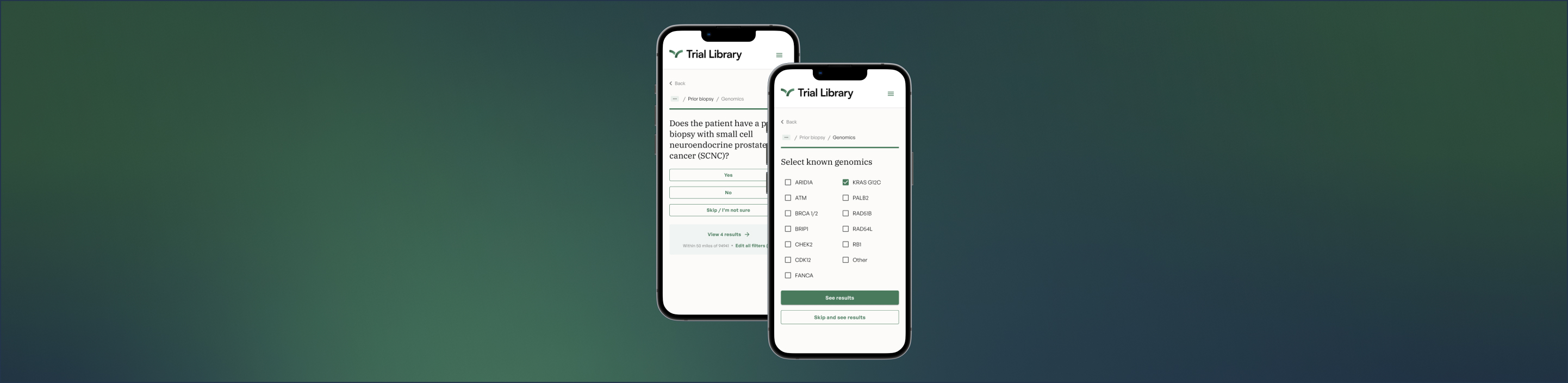
Trial Library is taking a different approach
We are introducing a new approach to the community-engaged research model. In speaking with many oncologists practicing in the community, trial sponsors, investigators, and patients, we heard repeatedly that there needs to be an end-to-end solution to motivate community oncologists to get engaged with referring patients to clinical trials at nearby trial sites. As we dug in deeper, we learned that community oncologists, who typically work at small to medium-sized physician owned practices, treat more than half of all patients with cancer. And most oncology practices, especially those in community settings, do not participate in clinical trials.
Identifying the reason for the lack of participation in clinical trials wasn’t difficult. To screen for a given clinical trial, a physician and staff must spend hours of effort combing through medical records. EMRs and other software tools fail to automatically do this work for most oncology trials. Not only is screening a practice for clinical trial candidates a lot of work but practices aren’t paid for any of their time or effort, so naturally, screening for trials is something that falls to the bottom of the list of priorities for community oncology practices.
We aim to change the lack of engagement in trials by creating a new set of tools for providers to get paid for their work in screening for trials and become true partners with investigators at trial sites. This solves what we are calling the “first mile problem” of clinical trial engagement. We couple this with our patient navigation support tools that solve the “last mile problem” in order to provide the complete solution this industry has lacked.